Do you wish you could make the leap to build your own study within an EDC solution? Perhaps you have relied on CROs or external developers and want more control, increased speed, or better cost effectiveness. Many of our customers come to OpenClinica specifically for these reasons.
Most electronic data capture systems are not designed for the average data manager to build studies. These systems often require a high level of technical expertise from specialized clinical programmers. On the other hand, systems that data managers may find easy to use are often over simplified, lacking the necessary capability and functionality to run a trial effectively. OpenClinica gives data managers the best of both worlds: exceptional capabilities and the ability for today’s clinical data managers to deploy them in their studies.
Let’s start by taking a look at some of the key ways the OpenClinica Study Designer delivers on this promise.
Building Study Events (e.g. Visits)
Events are the building blocks of your study’s architecture. This is where your protocol foundation is visualized, eCRFs are placed, and ultimately your study calendar is set. These building blocks can’t be set in concrete however. They must allow for flexibility during adaptive trials, during early phase setup, and any potential mid-study changes or protocol amendments down the road.
The following video shows how easy it is to create and modify study events:
Seamlessly engage patients
The Covid-19 pandemic underscores the need to be able to carry out trials more remotely, and as a result researchers are relying more heavily on ePRO, eCOA, and eConsent. These capabilities can be seamlessly added within the OpenClinica Study Designer. In fact, 90% of our clients are incorporating this digital capability within their studies now.
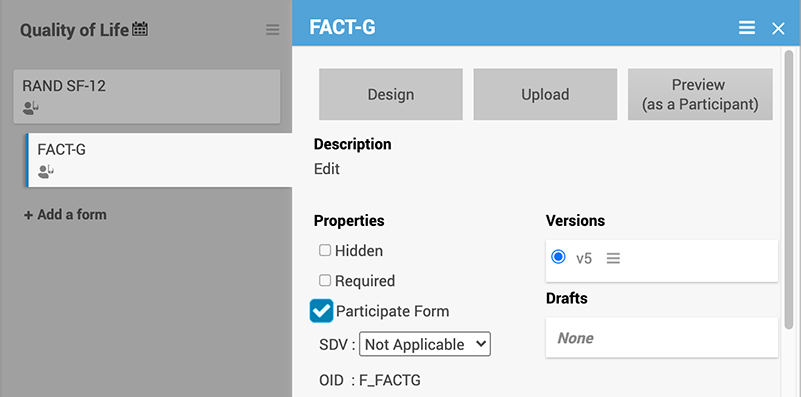
Select a single checkbox to enable an ePRO form.
Create ultra-smart eCRFs
The electronic case report form (eCRF) is the heartbeat of your study. OpenClinica eCRFs are incredibly capable and smart. Never ask the user for a value the system doesn’t already know (or can’t deduce). Real time edit checks and dynamic logic create a tactile and engaging experience. User forgot to hit the save button? No worries, OpenClinica forms automatically save your data. Whether a simple quality of life questionnaire or forms with edit checks, skip logic, and complex calculations, OpenClinica forms can have a tremendous impact on your study’s overall productivity.
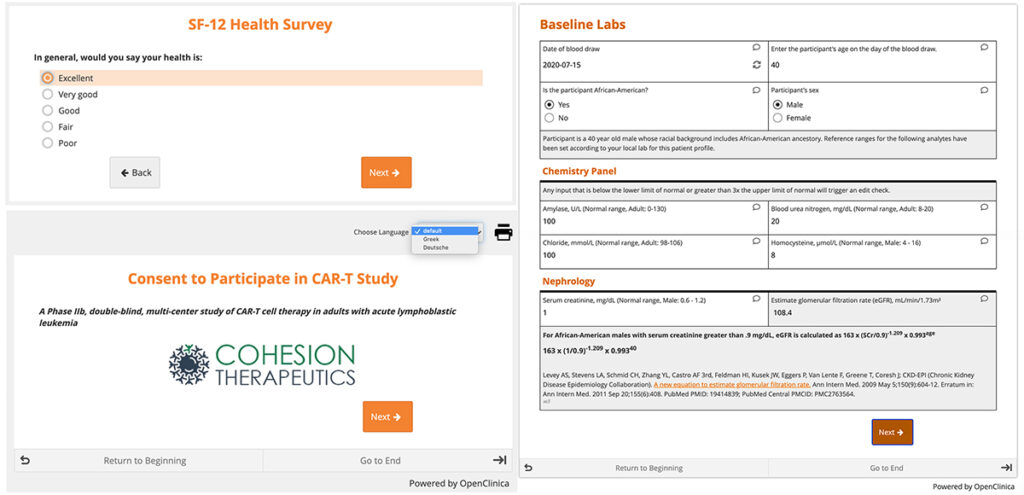
Check out our Ultimate eCRF Design Guide to see some OpenClinica forms in action. All the extraordinary things these forms can do can be set-up by a clinical data manager (you don’t need to be a programmer).
Stay tuned for a future post on the OpenClinica Form Designer and Form Library.
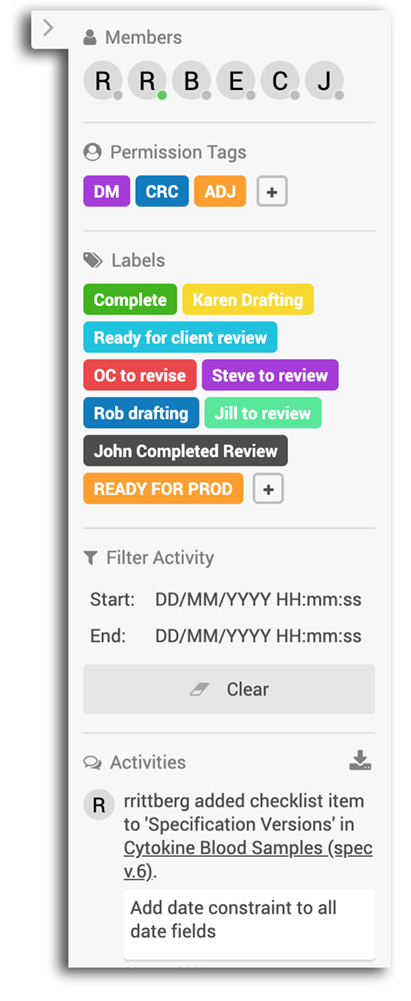
Collaborate with stakeholders
The pace of building a study is heavily impacted by the ability to collaborate and iterate with your team. With OpenClinica, you can avoid the countless emails and inefficient meetings which can frustrate your team and inflate the timeline unnecessarily.
OpenClinica’s Study Designer enables real-time and asynchronous review. Invite a colleague to review a form and post comments. See updates to the study design without having to refresh your screen. Use labels and checklists to track the review and approval process and keep everyone on the same page. Increase your velocity.
One-click publish
From the Study Designer, you only need a single click to publish your study to OpenClinica’s test or production RTE (run time environment).
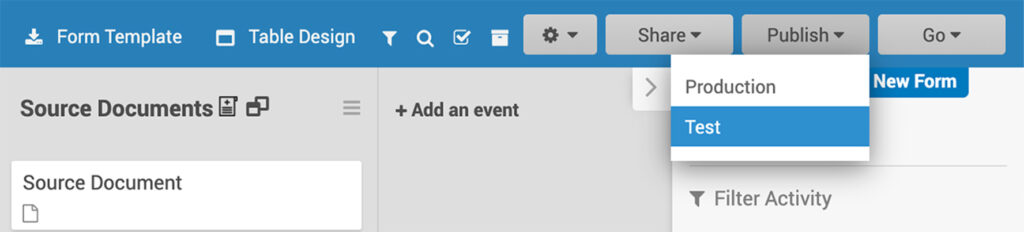

What you don’t see, running behind the scenes, is all of the automation, API calls, and modules associated with getting all your events, forms, definitions ready for go live. This is another example of powerful capabilities wrapped within a friendly, easy to use interface.
Publishing to your dedicated test environment, your team can perform all the training and testing required until you are ready to one-click publish to production. And don’t stress when the all too certain protocol amendments occur. You can make your study modifications quickly, whether event-based or form-based in the study designer. Rest assured, OpenClinica has fail-safe mechanisms for preserving study and data integrity.
Your stakeholders will be impressed at how you can turn around changes in hours, not not weeks.
No more long build times that bleed into study execution
No more advanced technical demands on data managers
No more wasted time in reviewing and publishing your study



